Can foreign companies make it into China’s Biopharmaceutical Market?
China has always been a hotbed for business opportunities, and the biopharmaceutical industry is no exception. With a population of over 1.3 billion and a rapidly growing middle class, there are plenty of potential customers for pharmaceutical products. However, foreign companies have often found it difficult to break into the China market.
In this blog post, we’ll take a closer look at the challenges and opportunities that foreign companies face when trying to do business in China’s biopharmaceutical market. We’ll also explore some strategies that you can use to maximize your chances of success in this tricky but lucrative market. So read on to learn more!
Cost-Effective Agency
KPI and Results focused. We are the most visible Marketing Agency for China. Not because of huge spending but because of our SMART Strategies. Let us help you with: E-Commerce, Search Engine Optimization, Advertising, Weibo, WeChat, WeChat Store & PR.

How Many Biopharma Companies Comprise the China Biopharmaceutical Market?
China has one of the largest biopharmaceutical markets in the world. Consisting of at least 400 biotech companies, the Chinese biopharmaceutical market is second only to that of the United States which still maintains a competitive advantage.
These biotech firms are a hotbed of innovation within healthcare and biotechnology. Among these firms are the likes of Sinopharm Group, one of the creators of the COVID-19 vaccine.
More players are entering the Chinese biomedical industry. The growth in participation is projected to bolster the country’s capital markets. The promotion of domestic participation is the result of the growing demand for access to quality healthcare and pharmaceuticals and medical products. Besides the clamor for innovative products like solid drugs, the design of government policies also plays a role in domestic biopharmaceutical product manufacturing.
What Is the Biggest Pharmaceutical Company in China?
When we think of the biggest biopharmaceutical company in China, it can mean one of two things. It can be in terms of the total market share of the Chinese pharmaceutical industry or it can be in terms of annual revenue.
It is crucial to make this distinction because market dominance through innovation of biopharmaceuticals and profiting thereof do not mean the same thing. Two companies can be credited with these feats in the Chinese market.
The biggest Chinese biopharma firms are:
Largest of local Biopharma players in Market Share: Sinopharm Group
As of May 2021, Sinopharm Group is the largest pharmaceutical company in China. Sinopharm Group accounts for the largest biopharmaceutical market share in China. Sinopharm Group has a market share worth at least $271.1 billion. This market share value is 10 times that of Shanghai Pharmaceuticals, another prominent player in the domestic market.  The market share that Sinopharm Group occupies represents the domination of the Chinese biopharmaceutical market. At the time of writing, more than 400 Chinese biotech companies make up the country’s increasingly competitive biopharma market.
The market share that Sinopharm Group occupies represents the domination of the Chinese biopharmaceutical market. At the time of writing, more than 400 Chinese biotech companies make up the country’s increasingly competitive biopharma market.
The Sinopharm Group’s market monopoly of the Chinese pharmaceutical market was not just possible in 2021. A look at its company background and 2020 revenue will reveal that the Chinese biotech firm also raked in sales amounting to $66.3 billion. Many experts have attributed Sinopharm Group’s success to both biopharmaceutical innovation in many areas and the COVID-19 pandemic.
Largest in Terms of Profit: Shanghai Pharmaceuticals and China Resources Pharmaceutical Group
Besides Sinopharm Group, other Chinese biopharmaceutical companies have enjoyed a large slice of the COVID-19 pie. Among these companies are Shanghai Pharmaceuticals. According to Statista, while Sinopharm Group occupied a large share of the Chinese market, Shanghai Pharmaceuticals topped the 2021 charts in revenue. Shanghai Pharmaceuticals brought in revenues of as much as $191.91 billion, making it the most profitable firm in the Chinese biopharma industry.

In regards to 2021 revenue, China Resources Pharmaceutical Group did not lag too far behind. Based on the same data, 2021 was the year China Resources Pharmaceutical Group made more than $175 billion. This makes the Hong Kong-based firm the second-largest biotech company in the Chinese pharmaceutical market.
Other biotechnology companies worth mentioning are Guangzhou Baiyunshan Pharmaceutical and Shenzhen Neptunus Bioengineering. These and at least nine other Chinese biotech companies top the Chinese market for pharmaceutical and medical products.
The Founding of the China Food and Drug Administration
The development or evolution of the Chinese biopharmaceutical industry can be traced back to the 1980s. It was during this time that policies were enacted to address the current healthcare issues of the times.
The implementation of healthcare policies required the presence of an overseeing body. The founding of an agency came to fruition more than a decade later with the founding of the State Food and Drug Administration in 1998.
The body oversaw the implementation of healthcare policies like the prioritization of primary healthcare. With many of the country’s primary healthcare targets met, there were still blind spots. In particular, the country lacked sectoral innovation coupled with a near-non-existent biopharma innovation ecosystem.
In other words, much work was necessary to incentivize biotechs in innovation. For this reason, the State Food and Drug Administration expanded its national healthcare strategy. The expansion included support for biopharma innovation players. Support came in the form of policies aimed at broadening access to markets
With the paradigm shift in its policies and capability requirements came the rebranding of the State Food and Drug Administration. In March 2013, the State Food and Drug Administration became the China Food and Drug Administration.
Innovation policies
Under the China Food and Drug Administration’s oversight, Business Research and Development and Innovation now operated under government policy that strengthened industry participants. By extension, the resulting biopharmaceutical policy evolution promoted access to quality healthcare made possible by the innovative product launches of Chinese biotech firms.
The Factors Behind the Rise of Chinese Innovations in Biotech and the Country’s Bio-industries
China has one of the largest biopharmaceutical and medical markets in the world, second only to the United States… it’s a hot spot. As more and more innovations take place within the mainland’s bio-industry bases like Wuhan and Shanghai, the biotech market grows at a rapid pace.
Industry experts attribute the boom of the industry to several factors. These factors are as follows:
Accessible Analytics
Data serves many biotechs in China. With open access to data, companies are better able to develop products that are both competitive and innovative. One example of an accessible source of data is GBI.
GBI is one of the most trusted healthcare analytics platforms in China. A GBI source is often reliable, as many industry experts will attest. Widely recognized in its value to the biomedical industry, GBI has grown into a massive healthcare analytics source with commercial capabilities.
More Incentives for Academia-to-Industry Innovations
The China Food and Drug Administration’s synthetic policies create the perfect environment for biotechnology innovation for broader healthcare ecosystems. The government favors and encourages innovation in all its forms, including those from the four walls of university campuses.
For this reason, China is a country where academia-level findings easily have commercialization capabilities. While some industry experts identify a gap between academia and industry, it continues to close.
An Aging Demographic
Innovations in drug discovery were not spearheaded just for the general population. It is worth noting that China is home to an aging population. At the time of writing, China’s elderly stands at 264 million. This is nearly a seventh of the country’s population.
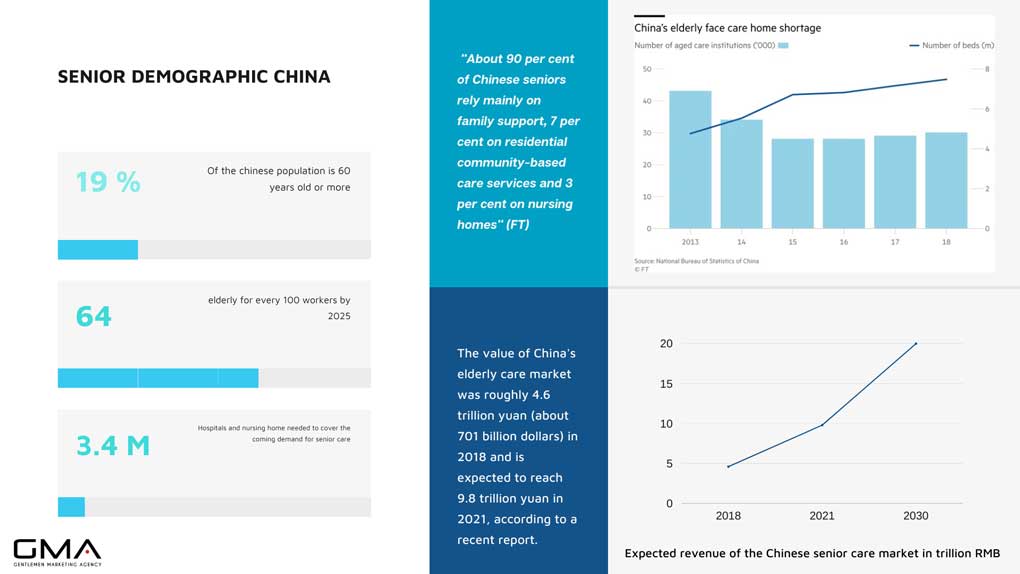
Not only is China home to a large elderly population. The country is also on the verge of a demographic time-bomb engendered by low birth rates and longer expectancies. According to the World Health Organization, China’s above-60 population will occupy 28% of the country’s population by 2040.
With a rapidly aging population comes the need for innovative drugs aimed at both the young and old. More biopharmaceutical technology sources mean broader public access to these drugs. Capitalizing on current market needs, many Chinese companies developed their biotech adeptness and agility, as well as their pharmaceutical marketing capabilities.
The Current COVID-19 Situation
COVID-19 has done much to reinforce the necessity for innovative drugs and other innovations in biotech in China and the world. Besides vaccines and innovative medicines for the virus, the pandemic gave birth to broader healthcare ecosystems.
During the early stages of the pandemic, the demand for other products like masks and personal protective equipment skyrocketed, placing pressure even on domestic supply chains and manufacturers. While many parts of the world are easing mask mandates, the demand remains high.
As well, many countries continue to work towards herd immunity status, adding to the demand for vaccines.
Can Foreign Companies Easily Break Into the Chinese Biopharma ecosystem?
With a biopharma market share that trumps that of European markets, the China biopharmaceutical market is a goal for any ambitious multinational company. Indeed, biotech is a strategic industry for such a company. One can simply look at multinational pharma companies such as AstraZeneca and Pfizer. However, a look at how these and other companies have penetrated the market is telling of how steep the odds are for foreign companies.
Access to markets can be tricky for foreign Pharmaceuticals & Medical Products companies
Many foreign companies have broken into the Chinese biotech industry through partnerships. Needless to say, these partnerships are with China’s major industry players. Owing to government policies and the drug regulation that regulate major stakeholdership by a foreign company, roles for foreign firms were limited. Often, these were in research and development.
Part of this has a lot to do with drug regulation and government policies. As mentioned earlier, both the China Food and Drugs Administration and the central government have designed policies to favor domestic industry participants and to ensure local drug quality standards. These policies make it difficult for foreign drug companies to enter the pharmaceutical market without Chinese partners.
In short, foreign companies can enter the market. However, resistance comes in the form of government policy. At most, foreign companies can break into the market as partners instead of major stakeholders.
When Did foreign drugs &Pharmaceutical Companies first enter China?
Foreign pharmaceutical companies looked Eastward in 1996. Many companies overseas entered the market, partnering with established Chinese biotech start-ups & companies. Such partnerships are key for domestic firms to stay in line with international standards
The nature of these partnerships was often investment-based, they are China Joint Venture. In fact, in 1996 alone, 40% of all state-owned biotechs were hotbeds of foreign investment from companies like Novartis, Glaxo, and Merck and Co. Also joining the investment roster at the time was Takeda Chemical, one of the largest manufacturers of biochemical products in the world.
Want to enter China Biopharma Market?
Contact us, we have solutions for you






